Other Targets
We are currently studying also many other transporters and protein targets and there results haven’t published yet. Herein, we list all the ongoing projects and the partners.
OCTs and multifunctional metformin derivatives
We have collaborated with Dr. Magdalena Markowicz-Piasecka, from Lodz Medical University, Poland since 2016. Main aim of our collaborative research has been in the development of metformin prodrugs and derivatives that would have greater anti-diabetic and anti-coagulant effects than metformin itself, but we have also studied the novel compounds as anti-cancer agents and as potential acetyl/butyrylcholinesterase inhibitors.
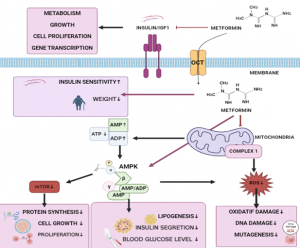
Recently, we have started to study the interactions of metformin and its derivatives with organic cation transporters 1-3 (SLC22A1-3), which we are slowly leveraging to othe SLC22 family members and to Multidrug and Toxin Extrusion (MATE; SLC47) family.
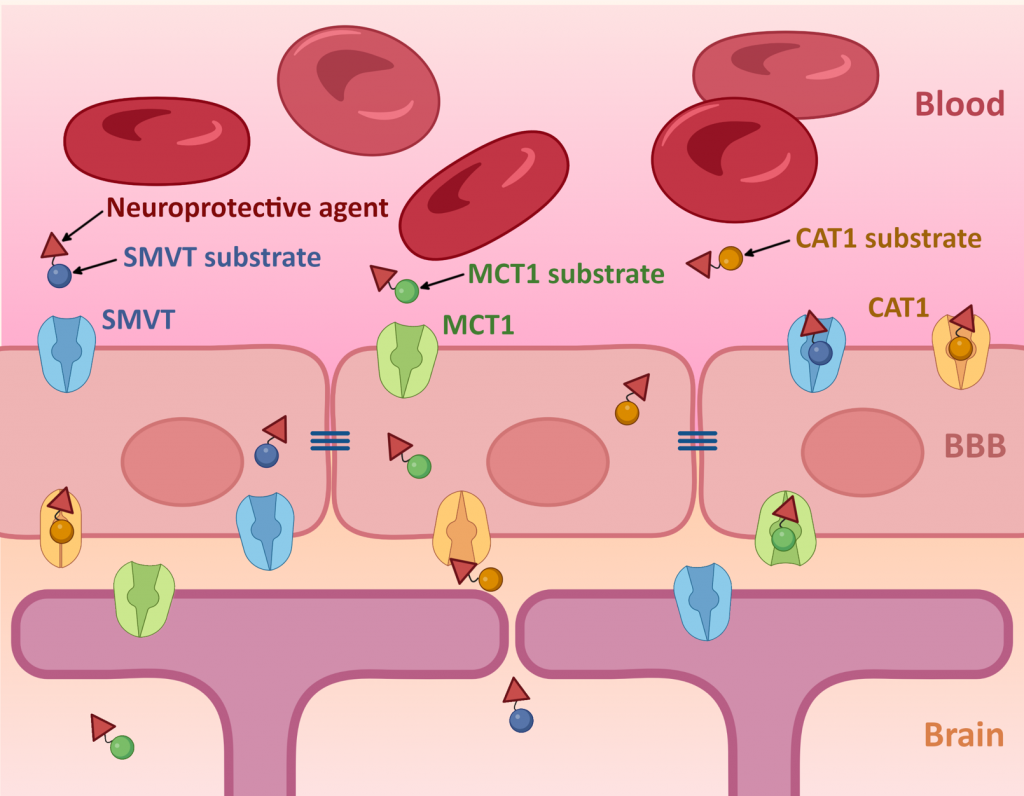
CAT1 & MCT1
Currently, in addition to LAT1 and OATPs, we are studying also cationic amino acid transporter 1 (CAT1, SLC7A1), monocarboxylate transporter 1 (MCT1, SLC16A1) and their possibilities to be utilized for brain- and intrabrain-targeted delivery of endogenous compounds in their novel transporter-utilizing prodrug forms. The computational drug design and studying the transport mechanisms of the designed substrates are carried out in a collaboration with Professor Antti Poso, UEF/Eberhard Karls University of Tübingen (Germany).
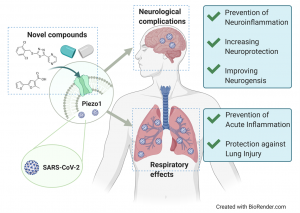
Novel agonists to modulate Piezo1 ion channel function
The involvement of Piezo ion channels in mechanosensitive transductions was awarded with a Nobel prize to David Julius and Ardem Patapoutian in 2021. Since 2019, we have collaborated with Professor Tarja Malm (UEF, A.I. Virtanen Institute; Neuroinflammation) to develop novel compounds that could function as a Piezo1 ion channel agonists. Prof. Malm’s group is shown that activation of Piezo1 increases microglial phagocytosis, reduces microglial pro-inflammatory activation, and enhances lysosomal activity, resulting in Aβ clearance both in human and mouse models of Alzheimer’s disease. On the other hand, Piezo1 is also expressed in the lungs and therefore, it is a potential multitarget druggable protein, by which, e.g., COVID-19-induced lung injuries accompanied by neurological complications could be treated.
Protein Phosphatase 2A (PP2A)
In collaboration, with Professor Timo Myöhänen (University of Helsinki) we are currently developing a novel activators of protein phosphatase 2A (PP2A) that could be utilized to treat neurodegenerative diseases with amberrant tau protein, so called tauopathies, such as Alzheimer’s disease (AD), Parkinson’s disease (PD) and Frontotemporal dementia (FTD). PP2A is the primary enzyme (phosphatase) to dephosphorylate or cleave the phosphate groups of hyperphosphorylated tau protein, and PP2A dysfunction has been linked to pathological hallmarks of neurodegenerative disorders. Therefore, modulation of PP2A function is highly interesting approach to treat several neurodegenerative diseases. This collaboration, that has been initiated in 2019-2020, involves also molecular modelling of the target protein, carried out by Dr. Maija Lahtela-Kakkonen (UEF, School of Pharmacy; Pharmaceutical Chemistry).
Placental Proteomics
Since 2021, we have collaborated with Professor Jaana Rysä (UEF, School of Pharmacy; Toxicology) to figure out changes in placental proteome and metabolome related to diabetes. Therefore, we aim to identify placental proteome and metabolites that could serve as predictive biomarkers for the wellbeing of the fetus and post-natal development as well as putative gender-specific molecular mechanisms linking diabetes with adverse outcomes of aberrant growth of the fetus.